
People walk in to get their COVID-19 vaccine at the Baldwin Hills Crenshaw Plaza past a sign that says "Johnson & Johnson vaccine only" in Los Angeles (Image via AP Photo/Damian Dovarganes, File)
After serious blood clots developed in six women, states like NC halted the use of Johnson & Johnson vaccines for now.
North Carolina joined a growing number of states pausing the use of the Johnson & Johnson vaccine Tuesday, after federal regulators launched an inquiry into a handful of “extremely rare” but serious blood clots.
“Our goal is that everyone receives a safe vaccine,” said NC DHHS Sec. Mandy Cohen, explaining her decision to halt the use of the J&J vaccine here.
The halt came after six women between 18 and 48 years-old developed cerebral venous sinus thrombosis, a very dangerous type of blood clots that can cause strokes. The women developed the rare condition within two weeks of receiving Johnson & Johnson vaccines, according to the CDC. One of the women died.
The six cases were among the 6.8 million people nationally who have received the one-shot dose of the J&J vaccine. That means only one out of more than 1,000,000 people experienced these serious problems.
“We’re recommending this pause while we work together to fully understand these events and also, so we can get information out to healthcare providers and vaccine recipients,” Dr. Janet Woodcock, the Food and Drug Administration’s acting commissioner, said on Tuesday. “Right now, I’d like to stress these events appear to be extremely rare.”
The halt may just last a few days, or weeks, as the CDC and FDA convene a panel of independent experts this week to determine how serious the issues are, and whether there should be additional advice about who should get the vaccine.
It does not affect the Moderna and Pfizer vaccines.
Both of those two-dose vaccines are being used here in North Carolina and around the country to thwart the spread of COVID-19, which has killed more than 562,000 people in the country since the start of the pandemic.
Rush to Vaccinate Hits Bump
The temporary shelving of the J&J vaccine, even if just a few days, is a major hiccup in the Biden Administration’s race to vaccinate as many people as possible. The J&J vaccine is one of only three to have emergency authorization for use in the United States. But because it can be stored more easily than the others and is only a single dose, instead of two, J&J was seen as a strategic tool to vaccinate harder-to-reach groups.
But health experts stressed that the pause is a sign that the nation’s system to monitor for adverse vaccination reactions works well.
“The system is working for us. Six events were detected, they’re concerning, out of almost 7 million doses of the Johnson and Johnson vaccine,” said Dr. David Wohl, an infectious disease physician at UNC Health in Chapel Hill. “We were able to see our federal colleagues recognize this, respond really quickly and make a call to action.”
The Moderna and Pfizer vaccines, which both received earlier approval and have been more widely used, have been used without any major issues.
“There has been no safety signal despite tens of millions of doses of both Moderna and Pfizer,” said Dr. Cameron Wolfe, a Duke University infectious disease doctor.
Wolfe said. “And our message, and my message to any patient I see is that we should continue to use those liberally while evaluating what’s going on here.”
What the J&J News Means for You
Here are some quick answers to questions you might have.
What happened?
Six women between 18 and 48 years old developed a serious but rare type of blood clot after getting their Johnson & Johnson vaccines. So far the clots have been reported only in women. The 6 cases are out of 6.8 million doses of the one-shot vaccine that have been given around the country.
Blood clots themselves aren’t that rare, with up to a half-million cases reported each year. But this particular type of blood clot, cerebral venous sinus thrombosis, is rarer and dangerous because it occurs in the brain and can cause strokes.
The CDC and FDA announced Tuesday morning they were recommending a pause in using Johnson & Johnon vaccines while they and others look into the incidents.
What should I do if I have a vaccination appointment?
People should keep their existing appointments with Pfzier and Moderna vaccines, according to the NC Department of Health and Human Services. Those two-shot vaccine regimens trigger the bodies’ immune system to protect against COVID-19 in a different way than the Johnson & Johnson vaccine.
“There have been no red flags” with those two vaccines, Dr. Anthony Fauci, the director of the National Institute of Allergy and Infectious Diseases, said Tuesday.
Those with J&J vaccines will have their appointment either canceled for now, or will be offered Pfizer or Moderna vaccines.
Vaccination clinics run by the UNC Health and Duke University systems, for example, have already switched to using Pfizer and Moderna vaccines and are urging people to keep their appointments.
Does this mean the J&J vaccine is not safe?
No, it doesn’t. The halt means that the handful of issues were detected and, because they were serious enough, prompted a pause for federal regulatory agencies to investigate and figure out the next steps. The CDC and FDA could end up changing its recommendations about who should get the vaccine, and how health care providers should monitor those at risk of developing the rare clots.
What should I do if I already got the J&J shot?
Many people report feeling aches and pains in the first two days after getting their vaccines, but these blood clots present differently, Cohen said.
If between 3 days and 3 weeks of getting your Johnson & Johnson vaccine you experience severe headaches, severe abdominal pain, difficulty breathing or leg pain, you should contact your healthcare provider and let them know you had the J&J vaccine, Cohen said.
Politics

Emergency rooms refused to treat pregnant women, leaving one to miscarry in a lobby restroom
By AMANDA SEITZ Associated Press WASHINGTON (AP) — One woman miscarried in the lobby restroom of a Texas emergency room as front desk staff refused...
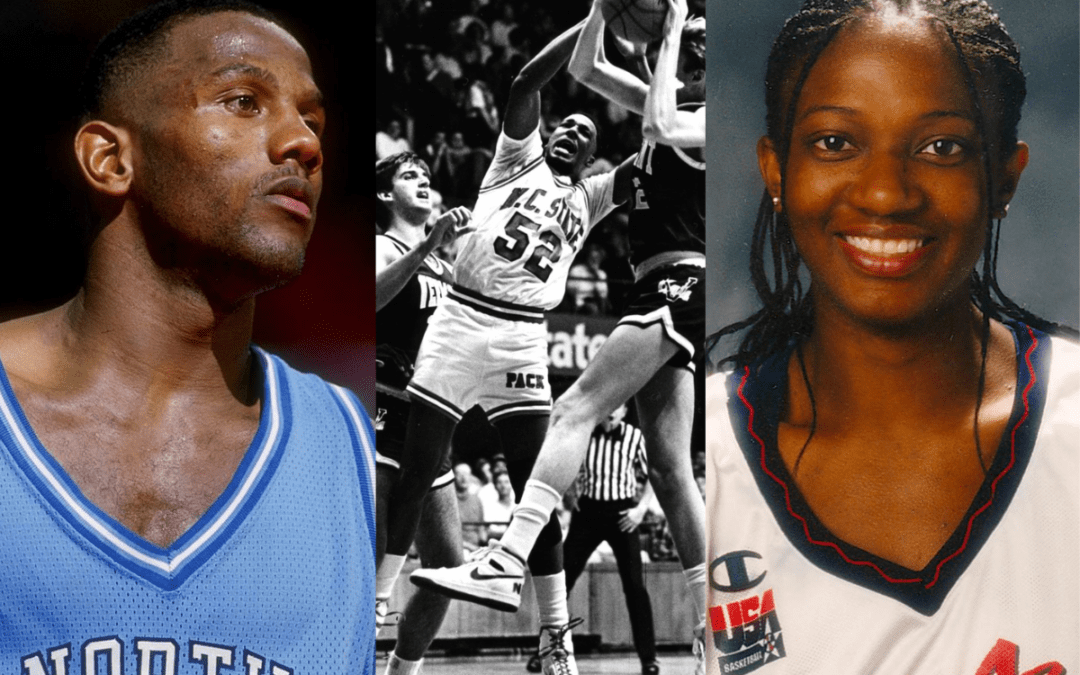
‘Unfit to be our next governor’: NC basketball greats speak out against Mark Robinson’s run for governor
NC has a long history of greatness on the basketball court. Now college basketball players from UNC, NC State, and Duke are wading into politics....
Local News

Good News Friday: It’s a good day to be a fan of the NC State Wolfpack
The men's and women's teams will compete for a national championship in college basketball this weekend. Plus: How to watch the solar eclipse, and...

The zodiac signs of 12 iconic women offer insight into their historic accomplishments
Zodiac signs can tell you a lot about someone’s personality. Whether they’re an earth, water, air, or fire sign, these 12 categories (which are...




